Clinical Research Certificate Course
Clinical Research Certificate Course - These include the basics of clinical trial design, regulatory requirements, and ethical. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical trials. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. Coursera offers a range of educational pathways in clinical research: If you are simply interested in properly understanding the published literature or if you are embarking on conducting your own research, this course is your first step. This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical research. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. In this course, you’ll learn about the more advanced elements of managing clinical trials. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. Clinical trials courses cover a variety of topics essential for understanding and conducting clinical research. These include the basics of clinical trial design, regulatory requirements, and ethical. If you are simply interested in properly understanding the published literature or if you are embarking on conducting your own research, this course is your first step. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical trials. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. In this course, you’ll learn about the more advanced elements of managing clinical trials. Professional certificates that can help you showcase your skills in clinical research settings. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. In the first part of the course, students will be introduced to terminology. Clinical trials courses cover a variety of topics essential for understanding and conducting clinical research. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. Clinical trials courses cover a variety of topics essential for understanding and conducting clinical research. In this course, you’ll learn about the more advanced elements of managing clinical trials. These include the basics of clinical trial design, regulatory requirements, and ethical.. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical trials. In the first part of the course, students will be introduced to terminology. This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical research. This. These include the basics of clinical trial design, regulatory requirements, and ethical. Transform you career with coursera's online clinical courses. From anticipating and planning for protocol events to conducting systematic reviews to synthesize. This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical research. If you are simply interested in properly. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. These include the basics of clinical trial design, regulatory requirements, and ethical. Coursera offers a range of educational pathways in clinical research: This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. In the first part of the course, students will be introduced. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. Professional certificates that can help you showcase your skills in clinical research settings. These include the basics of clinical trial design, regulatory requirements, and ethical. If you are simply interested. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. Professional certificates that can help you showcase your skills in clinical. Clinical trials courses cover a variety of topics essential for understanding and conducting clinical research. This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical research. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical trials. This course is designed to introduce you to the basic principles and practices of good clinical practice (gcp), which are essential for conducting clinical trials and ensuring the safety and. Clinical trials courses cover. These include the basics of clinical trial design, regulatory requirements, and ethical. Transform you career with coursera's online clinical courses. This course presents critical concepts and practical methods to support planning, collection, storage, and dissemination of data in clinical research. Professional certificates that can help you showcase your skills in clinical research settings. In the first part of the course,. Coursera offers a range of educational pathways in clinical research: Clinical trials courses cover a variety of topics essential for understanding and conducting clinical research. In this course, you’ll learn about the more advanced elements of managing clinical trials. The course will explain the basic principles for design of randomized clinical trials and how they should be reported. In the first part of the course, students will be introduced to terminology. These include the basics of clinical trial design, regulatory requirements, and ethical. Our course series on ich good clinical practice equips trial investigators and clinical researchers with the essential knowledge required to conduct reliable and ethical clinical trials. Transform you career with coursera's online clinical courses. Professional certificates that can help you showcase your skills in clinical research settings. If you are simply interested in properly understanding the published literature or if you are embarking on conducting your own research, this course is your first step.JLI Blog
Clinical Research Certification BioGrademy
Home 1 Global Pharma
3 months online certificate course in clinical research Global Pharma
Best Clinical Research Certificate Courses in India
why choose clinical research
Certificate course in " Clincal Research & Clinical Data Management
Clinical Research Certification Training. Graduate Training for
Clinical Research Training Course I Clinical Research Online Training I
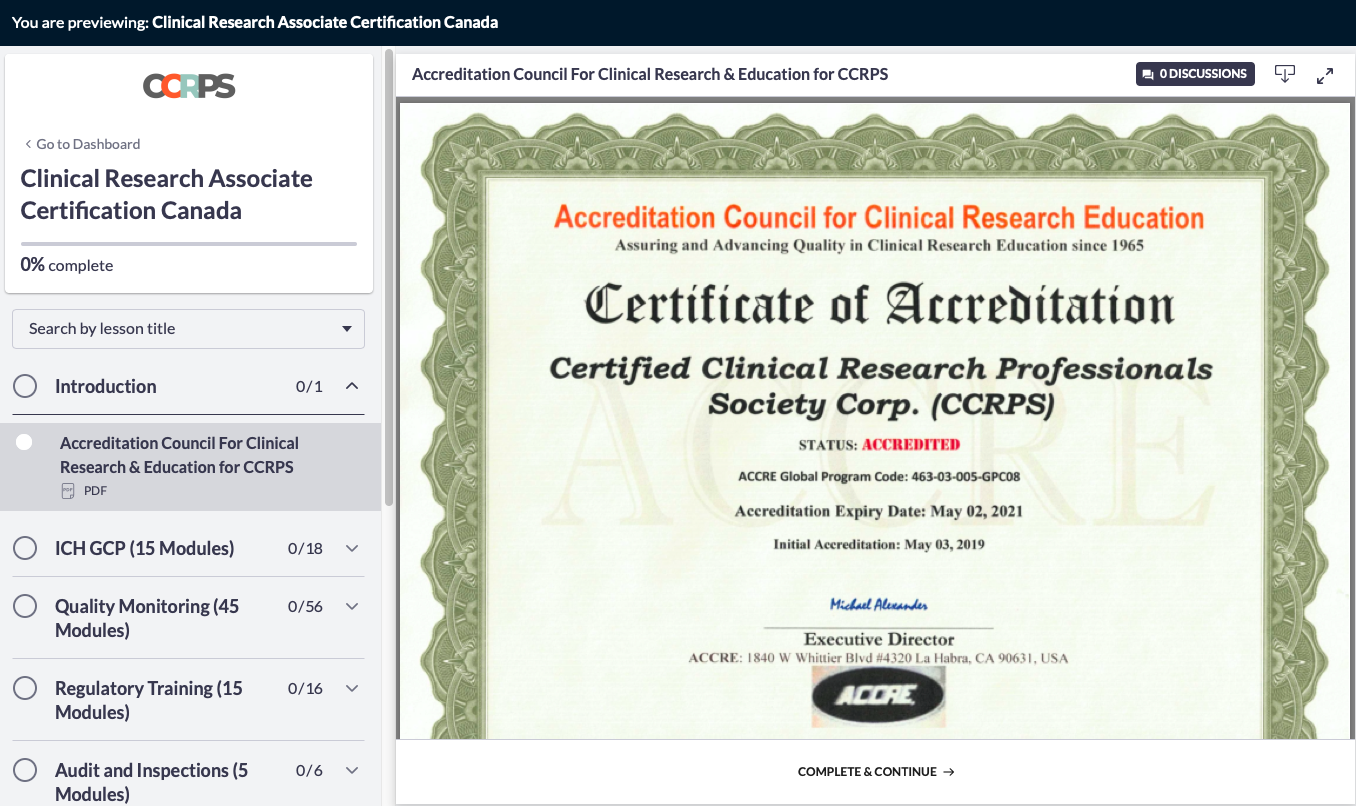
Clinical Research Associate Certification Canada Guide to A
This Course Is Designed To Introduce You To The Basic Principles And Practices Of Good Clinical Practice (Gcp), Which Are Essential For Conducting Clinical Trials And Ensuring The Safety And.
From Anticipating And Planning For Protocol Events To Conducting Systematic Reviews To Synthesize.
This Course Presents Critical Concepts And Practical Methods To Support Planning, Collection, Storage, And Dissemination Of Data In Clinical Research.
Related Post: